TECHNOLOGY
Advances in stem cell technology have greatly expanded the use of pluripotent stem cells and human-derived cells in various fields. For practical application, however, there remain several issues as to (1) method for producing functional cells and tissues, and (2) method for evaluating the function of those cells and tissues.
We develop unique and proprietary technologies for producing physiologically relevant cells and micro-physiological system (MPS) for functional evaluation of the cells, thereby contributing to a wide range of fields such as basic research, drug discovery, regenerative medicine, and cosmetics research.
Introduction: What is Three-dimensional Cell Culture?
Three-dimensional cell culture (3D cell culture) is a method of culturing cells in a three-dimensional environment. Compared to conventional two-dimensional cell culture (2D cell culture), where cells are cultured in a monolayer on a flat surface such as a cell culture plate, 3D cell culture creates a micro-environment closer to those in living organisms, thereby providing more physiologically relevant culture conditions.
Table 1.Comparison of 2D Cell Culture and 3D Cell Culture
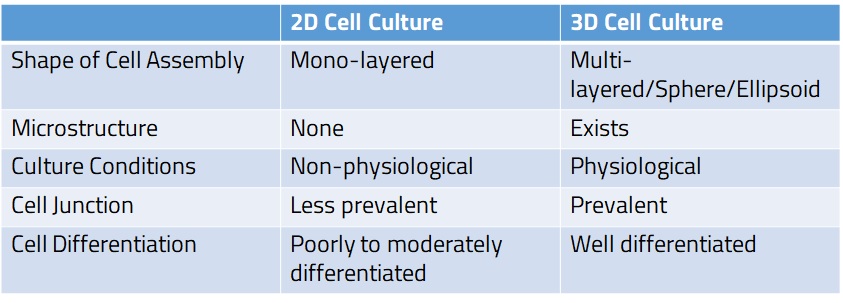
Ref. Miyamoto et al., Organ Biology 27: 37-52 (2020)
SCAD Technology of 3D Culture
We develop and use two major methods of 3D cell culture: (1) multi-layered cells using oriented fiber scaffold, and (2) self-assembly of cells in hydrogels.
1. Multi-layered cells using oriented fiber scaffold
Culturing cells on an oriented fiber scaffold yields a multilayered cell sheet where cells align along the fibers. When neurons are seeded on the fiber scaffold, the cell sheet with a thickness of 50 to 100 μm can be obtained with the neurons stacked on the fiber sheet. The multilayered neural cell sheets made with our technology have superior features to 2D culture: (1) acceleration of maturation of the cells by high-density 3D cell culture and (2) strong adhesion of the cells to the scaffold, which suppresses cell aggregation and allows stable culture for a long term.
(a) Oriented fiber scaffold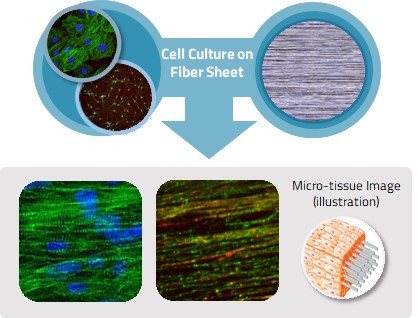
(b) Multi-layered neurons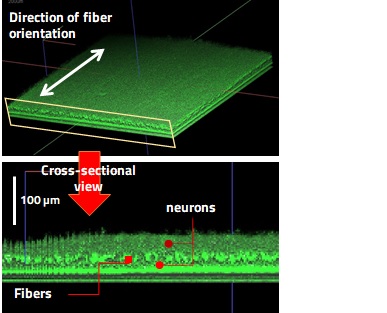
Fig. 1. 3D Cell Culture Using Oriented Fiber Sheet
2. Self-assembly of cells in hydrogels
Cell culture using a medium containing hydrogel controls cell migration and orientation, allowing the self-assembly of cells. By culturing skeletal myoblasts in a vessel having two posts, millimeter-sized skeletal muscle tissues, which cross-link the posts, are obtained (Fig. 2). The resulting skeletal muscle tissue contracts in response to electrical stimulation, which enables quantitative assessment of skeletal muscle contractility.
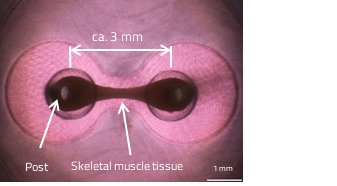
Fig. 2. 3D Cell Culture of Skeletal Muscle Tissue
“Cell Device”
We also develop unique cell culture vessels that can adapt to and be freely transferable between various containers and vessels. In addition, the cell culture vessels themselves are designed to have functions for assays. For example, cell culture on the device which is composed of an oriented fiber sheet and a small plastic frame that fits in a 96-well plate enable the cells to form a multi-layered cell sheet (Fig. 3a and 3b). Another example is a culture vessel for skeletal muscle tissue, which has two flexible posts for retaining skeletal muscle tissue and measurement of contractile forces (Fig. 3c).
We have named these culture vessels "cell devices" because they have two different functions, (1) cell culture and (2) functional evaluation, in a single vessel. We can flexibly design and prototype novel cell devices according to the purposes and target cells, taking advantages of microfabrication and plastic molding techniques.
(a) Fiber device

(b) Cross-sectional view of fiber device (illustration)
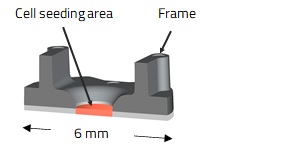
(c) Skeletal muscle cell device (illustration)
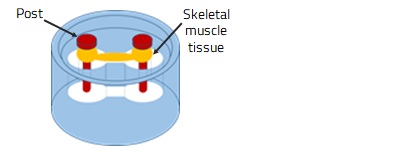
Fig. 3. Examples of Cell Device
Development of Methods for Functional Evaluation Using 3D Cell Culture
By utilizing our 3D culture technology based on the cell devices, we also develop novel methods for the functional evaluation of cells and tissues. We aim to create values by enabling quantitative assessment of the functions of target cells and tissues of live cells in vivo (e.g., nerve transmission, muscle contraction, etc.), which cannot be achieved by conventional methods.
1. Evaluation of electrophysiological properties of neurons
Because the primary role of neurons is to output and transmit electrical signals, electrophysiological measurement is crucial to understand the neural functions in both normal and abnormal conditions. In this regard, the measurement of extracellular potential of cells using multi-electrode array (MEA), one of the in vitro electrophysiological test methods, has received much attention in recent years.
MEA is a versatile technique for the evaluation of neural network activity, enabling the measurement of the extracellular potential of a cell population simultaneously and continuously. However, using neurons derived from iPS cells, the formation of neural networks usually requires a long culture period. It often fails to produce the desired results due to the aggregation of neurons that detach from the vessel during the culture. The multi-layered neuronal sheets made with our 3D culture technology resolve these issues and greatly enhance the usefulness of assessing the electrophysiological properties of neurons using MEA.
Measurement of extracellular potentials using MEA gives multiple analysis parameters such as the number of spikes, synchronous burst firing frequency, and synchronous burst duration. We have established multi-dimensional analysis by principal component analysis and hierarchical clustering of the MEA measurement analysis parameters.
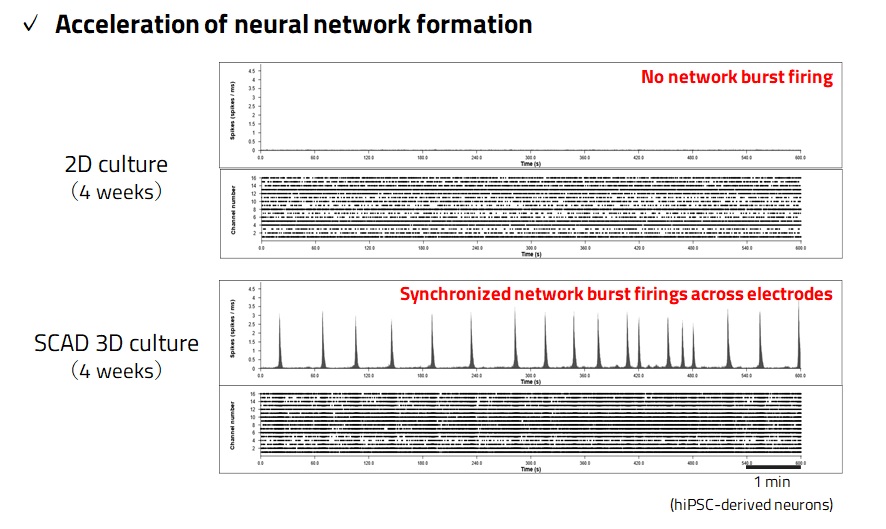
Fig. 4. Features of SCAD Neuron Device – Acceleration of Maturation by 3D Culture
2. Evaluation of contractility of skeletal muscle tissue
Skeletal muscle system plays a pivotal role in the physical functions of the human body. Though there have been many attempts to recreate the dysfunctions of contractility of skeletal muscles, conventional methods using cells or tissues have limitations from practical points of view. Firstly, 2D cell culture of the skeletal muscle on a flat surface is far from the environment in the human body, and myotube formation is significantly inhibited. Secondly, analysis of expression levels of genes or proteins, which are widely used techniques for evaluation of cells and tissues, have only limited predictability on the contractility of skeletal muscles.
Our 3D cell culture technology produces skeletal muscle tissues that contract in response to electrical stimulation. We also developed an in vitro assay for quantitative evaluation of contractility of skeletal muscle derived from mouse C2C12 cells. We confirmed that the assay could be used as a model of muscle atrophy induced by steroids. Treatment of the skeletal muscle tissue with dexamethasone caused a reduction of muscle contractility, which was rescued by the addition of IGF-1 (Fig. 5). Application to human cells is now underway.
(a) Muscle contractility at 48h after drug administration
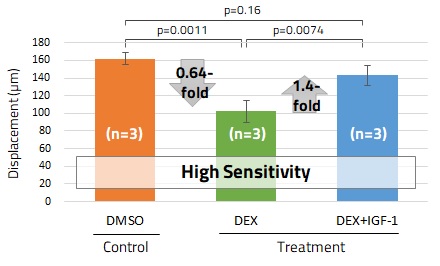
(b) Time-course change of muscle contractility
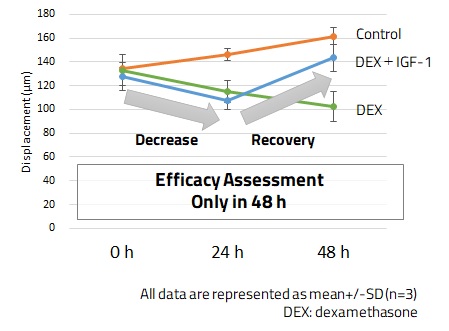
Fig. 5. Application of SCAD Skeletal Muscle Device – Evaluation of Muscle Contractility Using Mouse C2C12 Cells
3. In vitro model of neuromuscular junction
In vivo, skeletal muscle contraction is triggered by an action potential arriving at the neuromuscular junction where the receptors on muscle cells receive neurotransmitters released from motor neuron terminals. Disorders of the neuromuscular junction cause muscle weakness and atrophy, leading to age-related muscle loss or muscular dystrophy. In more severe cases, the loss of motor function cause life-threatening damage to the respiratory muscles, which is a most serious symptom of amyotrophic lateral sclerosis (ALS). We are also working on neuromuscular junction models by utilizing our technologies related to neurons, skeletal muscles, and MPS.
Patient-derived iPS Cells and their Application to Disease Modeling
Studies on elucidating the causes of diseases and novel therapies using iPS cells (disease-specific iPS cells) generated from patients' somatic cells or differentiated cells is making great progress. In the field of neuromuscular diseases, where there are many hereditary and intractable diseases, the use of disease-specific iPS cells is of great significance.
We are currently working on developing disease models for Charcot-Marie-Tooth disease (CMT). CMT are progressive neuromuscular disorders characterized by degeneration of the peripheral nerves leading to muscle atrophy and weakness in the distal limbs. Though CMT are the most frequent inherited diseases of the peripheral neuropathies with a prevalence of 1/1500 to 1/10000 worldwide, no drug is currently available. To facilitate and expedite the development of therapeutic approach to CMT, patient-derived disease models that recapitulates CMT phenotype are desperately needed.
In November 2019, we started a joint research project with Kyoto Prefectural University of Medicine. We are generating several iPS cell lines derived from multi-types of CMT patients to develop the disease models for drug discovery.
Fig. 6. Example of patient-derived iPS cells
